The world is evolving very rapidly and in the field of vaccines even more so this year 2020. Today there are several candidate vaccines to fight the sars-Covs-2 pandemic that has led to Covid-19 disease. Vaccines under development include Moderna, Oxford/AstraZeneca,Pfizer/BioNTech, Johnson & Johnson, Novavax and Sinovac.
This week we talked about the Oxford vaccine that is about to be approved for use in humans. The pharmaceutical company Astrazeneca, Oxford Biomedica and the University of Oxford have worked together for this vaccine. This article discusses that vaccine and how it was created.
Background
Astrazeneca is a global and innovative biopharmaceutical company focused on the discovery, development and marketing of prescription drugs.
Oxford Biomedica is a cell and genomic therapy company specializing in the development of gene-based medicines. In April 2020, Oxford BioMedica announced that the company had joined a consortium led by the Jenner Institute of the University of Oxfordfor the development and manufacture of a potential vaccine forCOVID-19. This is one of many vaccine candidates currently under development.
Cómo funciona la vacuna
The Oxford University vaccine trial can be explained as a copy-paste mechanism. It is explained in Fig. 1..
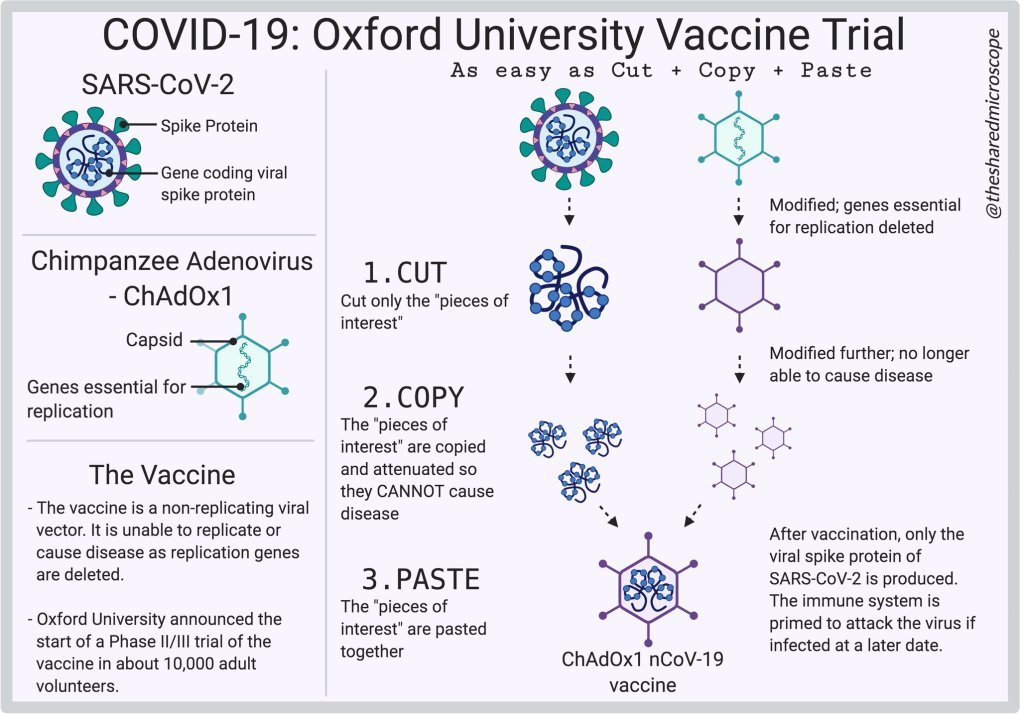
The process of creating this vaccine is part of two viruses: Sars-CoV-2 and ChAdOx1 virus (a chimpanzee adenovirus). The process is based on CUTTING only the parts that are of interest. COPY the pieces of interest and dim so as not to cause the disease. Finally, the pieces of interest are GLUED together. That is, they are assembled into a new virus called ChAdOx1 nCov-19. This particle is the vaccine itself and is called AZD1222, Fig. 2.

Once the vaccine is given, the body reacts to that viral particle and generates antibodies against it. But the big difference is that this new genetically engineered virus does not contain the spies that are the key in the process of adhesion to the cell membrane of the cell through the ACE2 receptor, see Fig. 3.

This ACE2 receptor is known to be found in various tissues of the human body that can be seen in Fig. 4. It is precisely these tissues that most severely affect Covid-19 disease. Therefore, there is clinical evidence that the Sars-Cov-2 virus is found in them.

Keys to the AstraZeneca vaccine announcement
There are several highlights to highlight in AstraZeneca’s announcement:
- Two different forms of dosage demonstrated efficacy and one showed a better profile, a higher success rate.
- No hospitalizations or severe cases of COVID-19 in participants treated with AZD1222.
One form of dosage (in a total of 2,741 cases) showed 90% vaccine efficacy when AZD1222 was administered as a half dose followed by a full dose at least one month apart, and another dosing regimen (a total of 8,895) showed 62% efficacy when two full doses were administered. dosages at least one month apart. AstraZeneca will now immediately prepare all necessary regulatory documentation of data to be submitted to authorities around the world that have an established framework for conditional or advance approval of this vaccine.
AZD1222 was jointly invented by the University of Oxford and its spin-off company, Vaccitech. It uses a poorly replicated chimpanzee viral vector based on a weakened version of a common cold virus (adenovirus) that causes infections in chimpanzees and contains the genetic material of sarS-CoV-2 virus peak protein. A viral vector functions as a vehicle for the introduction of a virus into a cell. After vaccination, peak surface protein is produced. Prepares the immune system to attack the SARS-CoV-2 virus if the body is exposed to the virulent agent.
Video de promoción
This is a video provided by Astrazeneca about how the vaccine is performed.
Summary
Therefore, we split two viruses, Sars-Cov-2 and a chimpanzee adenovirus. We know that the virulence of the Covid-19 virus is caused by its spies, its “antennae”. A new virus is built that on the one hand has the adenovirus capsule (which is not virulent) and the genetic material of the Sars-Cov-2 virus that are introduced into that capsule. We need you to have genetic material from the Sars-Cov-2 virus because our body has to be able to detect that antigen (foreign material) and create antibodies against it.
This new virus is not able to adhere to the cell membrane of the cell, since it does not have the proteins of the spies that allow that binding and therefore do not reproduce in the cell.
This process is done through a simple copy-paste process. However, to say that in biology there is nothing easy to do and what has been achieved has been based on work and work and work. Trial and error, trial and error. The company explains it that way, that even an anti-vaccine can understand it, but as I say it is the results of thousands of hours of work and dedication.
In the next few days I’ll upload more articles talking about how the other vaccines work. I hope it’s interesting to you. Remember that this information is online, I have searched the official source, not on a third-party page, and it is the interpretation that a biologist like me makes. I may be wrong, but in science there is nothing 100% safe and as a scientist you have to have an open mind to recognize the progress that is taking place and never stand still.
Sources / bibliography
- https://en.wikipedia.org/wiki/Oxford_BioMedica
- https://www.astrazeneca.es/nuestracompania.html
- https://www.astrazeneca.es/medios/notas-prensa/2020/azd_1222_vaccine_met_primary_efficacy_endpoint_in_preventing_covid19.html
- https://www.astrazeneca.es/medios/notas-prensa.html
- https://fancycomma.com/2020/07/02/all-about-the-oxford-astrazeneca-covid19-vaccine/
- https://www.jenner.ac.uk/
- https://www.oxb.com/news-media/press-release/oxford-biomedica-notes-astrazeneca-s-azd1222-met-primary-efficacy-endpoint
- https://www.instagram.com/thesharedmicroscope/
- Ilustrations made using BioRender.com








